A) release hydrogen ions when dissolved in a liquid
B) feel slippery when touched
C) taste bitter
D) release hydroxide ions when dissolved in a liquid
E) have a pH reading above 7.0
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The pH of pure water is ________ because ________.
A) 7.0; water dissociates an equal number of H+ ions and OH- ions
B) 14.0; water dissociates and more OH- ions are formed because there are more hydrogen atoms in water
C) 1.0; water dissociates and more H+ ions are formed since hydrogen is smaller and can separate from the oxygen easily
D) 7.0; there are no ions formed in pure water
E) acidic; there are more H+ ions than OH- ions present
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
After drinking a great deal of coffee pH 5) , a human's blood buffering system would need to ________ as the coffee was digested to lower the level of acid present in the blood stream.
A) release OH-
B) take up H+
C) release H+
D) take up OH-
E) release OH- and take up H+
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A covalent bond occurs when
A) protons are transferred from one atom to another.
B) neutrons are shared between two atoms to form an isotope.
C) electrons are shared between two atoms to complete their octets.
D) the hydrogen of one water molecule is attracted to the oxygen of another water molecule.
E) electrons are transferred from one atom to another.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of neutrons present in the nucleus of an average atom of any given element is best estimated by
A) adding the number of electrons and protons together.
B) subtracting the number of electrons from the number of protons.
C) adding the mass number to the number of electrons.
D) subtracting the number of protons from the mass number.
E) adding the atomic number and atomic mass together.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elements differ from each other in their
A) physical properties only.
B) atomic number only.
C) type of subatomic particles.
D) physical properties and atomic number.
E) type of electrons.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a compound?
A) H2O
B) O- 2-
C) NaCl
D) CO2
E) MgCl2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which property of water would help to account for how an individual who is exercising and producing excessive heat can maintain a constant body temperature?
A) Water has high heat capacity.
B) Water is less dense than ice.
C) Water is a good solvent.
D) Water is cohesive.
E) Water molecules form by covalent bonding.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Radioactive isotopes are useful in biological studies because
A) an organism will take in a molecule with the isotope and use it normally, but the radioactive decay can be detected.
B) an organism will take in a molecule with the isotope but will only use it in a few specific reactions, not the normal ones.
C) an organism will take in the molecule with the isotope and then remove the isotope by sending it through the excretory system, while replacing the isotope with a normal atom.
D) they are easily visible and normal atoms are not.
E) they are easy and inexpensive to use in studies.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A water molecule, as shown here, is polar because of 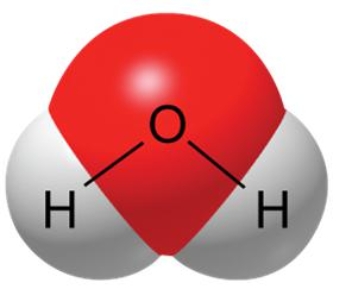
A) the transfer of electrons.
B) unequal sharing of electrons.
C) its ability to freeze.
D) its hydrogen bonds.
E) the negative charge of the molecule.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cola has a pH of 3.5. This means that it has an excess of _______ ions and would be called an) ________.
A) H+; acid
B) OH-; acid
C) H+; base
D) OH-; base
E) H+; neutral solution
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom with a neutral charge has
A) equal numbers of neutrons and electrons.
B) more neutrons making it more neutral.
C) the same number of protons and neutrons.
D) equal numbers of protons and electrons.
E) more protons than it does electrons.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not one of the most common elements in living things?
A) oxygen
B) carbon
C) calcium
D) iron
E) nitrogen
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom's valence electron shell
A) is filled when it has three electrons.
B) determines its chemical reactivity.
C) determines its atomic mass.
D) is filled with positively charged particles.
E) is filled identically for every element.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a way in which chemical bonds can be formed?
A) sharing electrons
B) losing electrons
C) splitting electrons
D) gaining electrons
E) attracting opposite charges
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 55 of 55
Related Exams