Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following properties is least characteristic of typical ionic compounds?
A) high melting point
B) high boiling point
C) brittleness
D) poor electrical conductor when solid
E) poor electrical conductor when molten
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For which of the following elements (in their normal, stable, forms) would it be correct to describe the bonding as involving "electron pooling"?
A) hydrogen
B) helium
C) sulfur
D) iodine
E) aluminum
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Most of the copper sold in major metal markets is highly purified, typically to 99.99%. Why is this?
Correct Answer

verified
The main use of copper is as a...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
In not more than three sentences, describe the electron arrangement responsible for bonding in Cl2 molecules.
Correct Answer

verified
This is covalent bonding. The chlorine a...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Acetone can be easily converted to isopropyl alcohol by addition of hydrogen to the carbon-oxygen double bond. Calculate the enthalpy of reaction using the bond energies given. 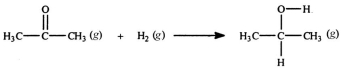
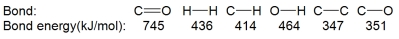
A) -484 kJ
B) -366 kJ
C) -48 kJ
D) +48 kJ
E) +366 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct formula for a compound formed from calcium and chlorine.
A) CaCl
B) CaCl2
C) Ca2Cl
D) Ca2Cl2
E) CaCl3
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Using appropriate, real examples to illustrate your answer, describe the correlation between bond energy and bond length for a series of single bonds.
Correct Answer

verified
The bonds between carbon, C, and the hal...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
The lattice energy of large ions is greater in magnitude than that of small ions of the same charge.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The stronger the bonds in a fuel, the more energy it will yield.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a covalent compound?
A) Na2O
B) CaCl2
C) Cl2O
D) CsCl
E) Al2O3
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogenation of double and triple bonds is an important industrial process. Calculate (in kJ) the standard enthalpy change ?H for the hydrogenation of ethyne (acetylene) to ethane. 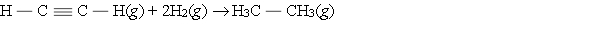
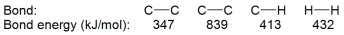
A) -296 kJ
B) -51 kJ
C) 51 kJ
D) 296 kJ
E) 381 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange oxygen, sulfur, calcium, rubidium and potassium in order of decreasing electronegativity.
A) O > S > Ca > K > Rb
B) O > S > Ca > Rb > K
C) O > S > Rb > K > Ca
D) O > S > Rb > Ca > K
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 73 of 73
Related Exams